
So let’s move on to think about a molecular crystalline solid and so what do I mean by a molecular crystalline solid, in this case the individual components aren’t atoms but they’re molecules and so this is an example of ice this is one crystal structure that water model molecules can take. In other cases we might be talking about covalent bonding or metallic bonding but the point is that this is very strong primary bonding so bond enthalpies can be quite strong between individual atoms. So see, there’s things like metals or ionic solids the individual components of that crystal structure are atoms so in this case this is an ionic solid so there might be some cations and some anions but the individual components are atoms and the bonding between the principal components is strong primary bonding and so in this case that might be ionic bonding. So in order to get started, let’s think about how we describe these different systems before An atomic crystalline solids. We are gonna basically focus on some definitions about what are liquid crystals were – some of the different kinds of liquid crystals and we’re going to talk about short and long range order in these systems. So we’re going to start talking about what these materials are. So that might seem like a little bit of an academic and not an interesting thing at all until you remember that a lot of technology is based on liquid crystals so a liquid crystal display is basically a something like a monitor or TV where we use a thin film of liquid crystal materials to turn on and off each pixel and we do that specifically by changing their internal structure, changing how they’re oriented so that it affects how they interact with polarized light. 
Specifically they can change the polarization of light and so this is basically an image of shining polarized light through a film of liquid crystals and you can see it some parts the light is basically rotated and slowed, some part of the light is cancelled out because it’s rotated and it’s rotated an angle that’s cancelled out by the polarizer that is between the film and where we’re looking at it. Liquid crystals are particularly important because they interact with light in interesting important ways. To help show this three-dimensional shape even more accurately, we can rely on space-filling models as well as ball-and-stick models.Today we’re going to talk about things that are not quite crystalline but definitely not amorphous as well and we’re a start off by talking about liquid crystals. We will discuss the significance of these electrons at the end of this section. The two dots above nitrogen indicate a lone pair of electrons that are not involved in any covalent bond. 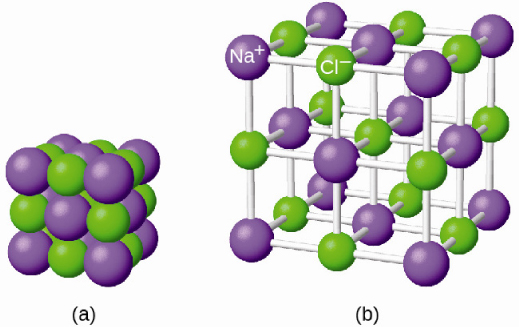
However, in the more detailed structural formula on the right, we have a dashed line to indicate that the rightmost hydrogen atom is sitting behind the plane of the screen, while the bold wedge indicates that the center hydrogen is sitting out in front of the plane of the screen. In the structural formula to the left, we are only seeing a two-dimensional approximation of this molecule. Keep in mind, however, that atoms and molecules, just like everything else in the universe, exist in three dimensions-they have length and width, as well as depth. From both of these structural formulas, we can see that the central nitrogen atom is connected to each hydrogen atom by a single covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
